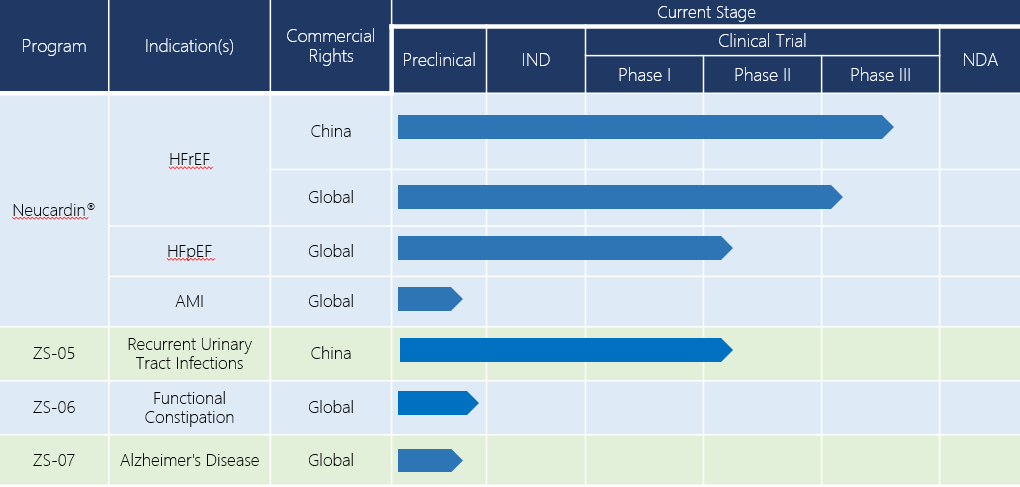
ZS-07
Alzheimer's disease is a kind of onset conceals the progressive development of the nervous system degenerative disease, clinical go up to memory impairment, aphasia, disuse, agnosia, depending on the spatial skills damage, executive dysfunction, and show the characteristics of the personality and behavior change and so on comprehensive dementia, pathogenesis is not fully elucidated.
Alzheimer's disease is the most common form of senile dementia and is among the leading causes of death in developed countries. There were an estimated 54 million people with Alzheimer's globally in 2018, with a compound annual growth rate of 3.0% between 2014 and 2018. Alzheimer's disease is closely related to the proportion of the elderly population over 65 years old. With the increasing trend of population aging, the number of Alzheimer's disease patients is expected to increase further in the future, reaching 63.3 million in 2023 and 80.6 million in 2030. The cost of treatment and care for Alzheimer's disease is already placing a heavy burden on society, with global health spending on Alzheimer's and other dementias reaching $1 trillion in 2019 and expected to double by 2030.
Glucose metabolism plays a significant role in the central nervous system. Unlike other organs, the brain depends entirely on the energy provided by glucose metabolism for neuronal activity, and an insufficient supply of blood glucose can cause neurological damage. The level of glucose metabolism is closely associated with cerebrovascular disease, amyloid β production and clearance, and Tau phosphorylation, indicating that abnormal glucose metabolism is closely related to the pathogenesis of Alzheimer's disease (AD). Zensun has therefore proposed a new strategy to improve brain energy metabolism in order to prevent and treat AD. We have generated ZS-07 as a multi-component synergistic combination of new therapeutic drugs affecting a variety of targets of brain energy metabolism.
Preclinical studies have shown that ZS-07 is a potential candidate for the treatment of AD., as it improves memory and autonomic activity, and decreases anxiety in animal models. The safety of various components of ZS-07 has been demonstrated over years of medical practice.